Research Overview
 Tooth loss, masticatory dysfunctions, swallowing difficulty, and pain states such as temporomandibular disorders, neuralgia and neuropathy, have devastating effects on human quality of life. Difficulty in swallowing, which is prevalent in 72%-87% of patients with Parkinson’s disease and 37%-78% in stroke, predispose patients to aspiration pneumonia that may lead to cardiorespiratory failure. Many age-related oral health problems have also been associated with Alzheimer’s disease and age- related dementias. Despite the importance of orofacial functions and the prevalence of disorders and dysfunctions affecting the feeding system, the cortical and biomechanical control processes that underlie orofacial functions are widely unknown. This represents an important problem because until they are understood, the cortical mechanisms underlying pathological aging and other diseases affecting this system remain largely incomprehensible, thus, hampering our ability to develop appropriate diagnostic tools, therapies and treatments.
Tooth loss, masticatory dysfunctions, swallowing difficulty, and pain states such as temporomandibular disorders, neuralgia and neuropathy, have devastating effects on human quality of life. Difficulty in swallowing, which is prevalent in 72%-87% of patients with Parkinson’s disease and 37%-78% in stroke, predispose patients to aspiration pneumonia that may lead to cardiorespiratory failure. Many age-related oral health problems have also been associated with Alzheimer’s disease and age- related dementias. Despite the importance of orofacial functions and the prevalence of disorders and dysfunctions affecting the feeding system, the cortical and biomechanical control processes that underlie orofacial functions are widely unknown. This represents an important problem because until they are understood, the cortical mechanisms underlying pathological aging and other diseases affecting this system remain largely incomprehensible, thus, hampering our ability to develop appropriate diagnostic tools, therapies and treatments.
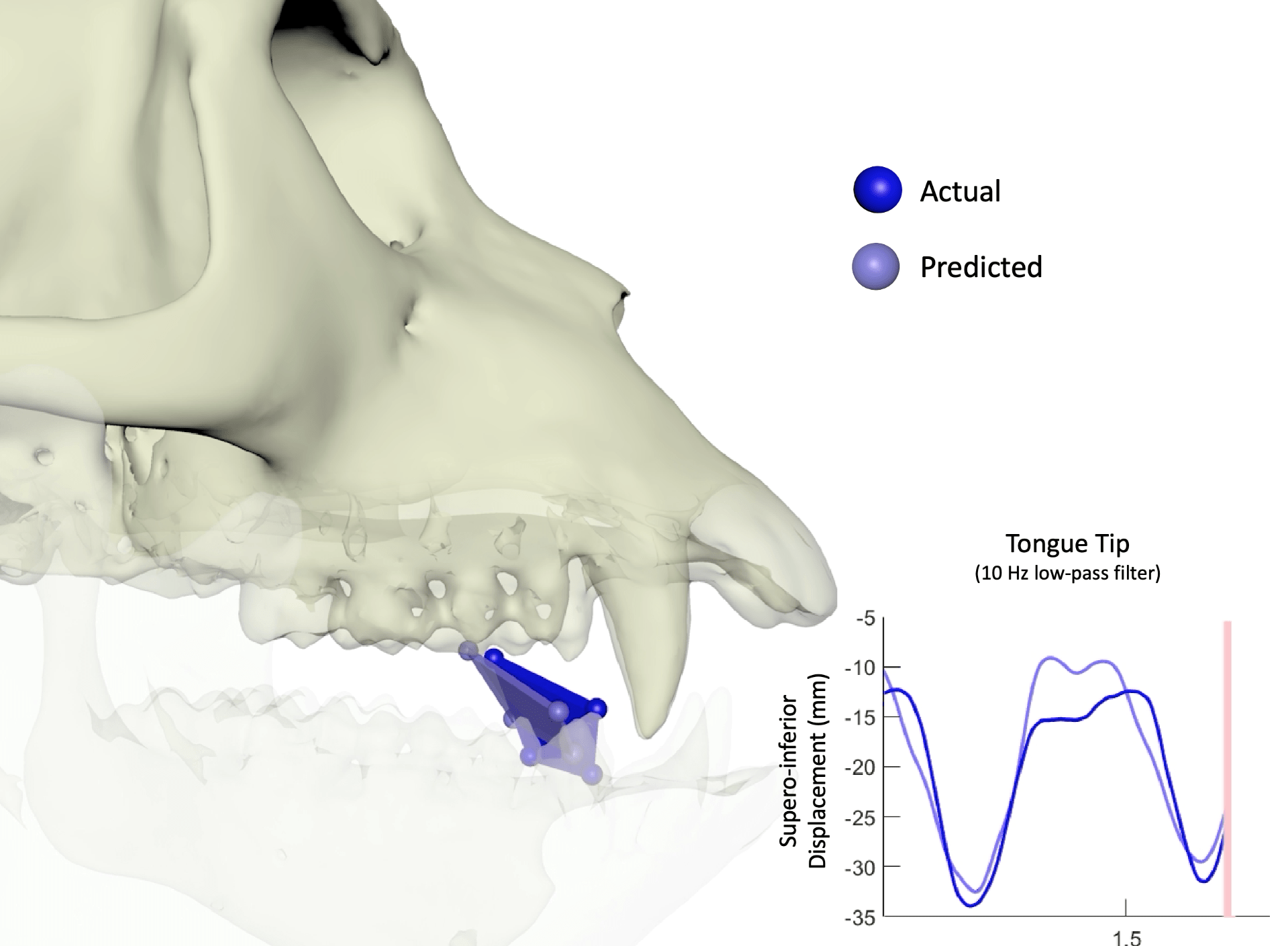
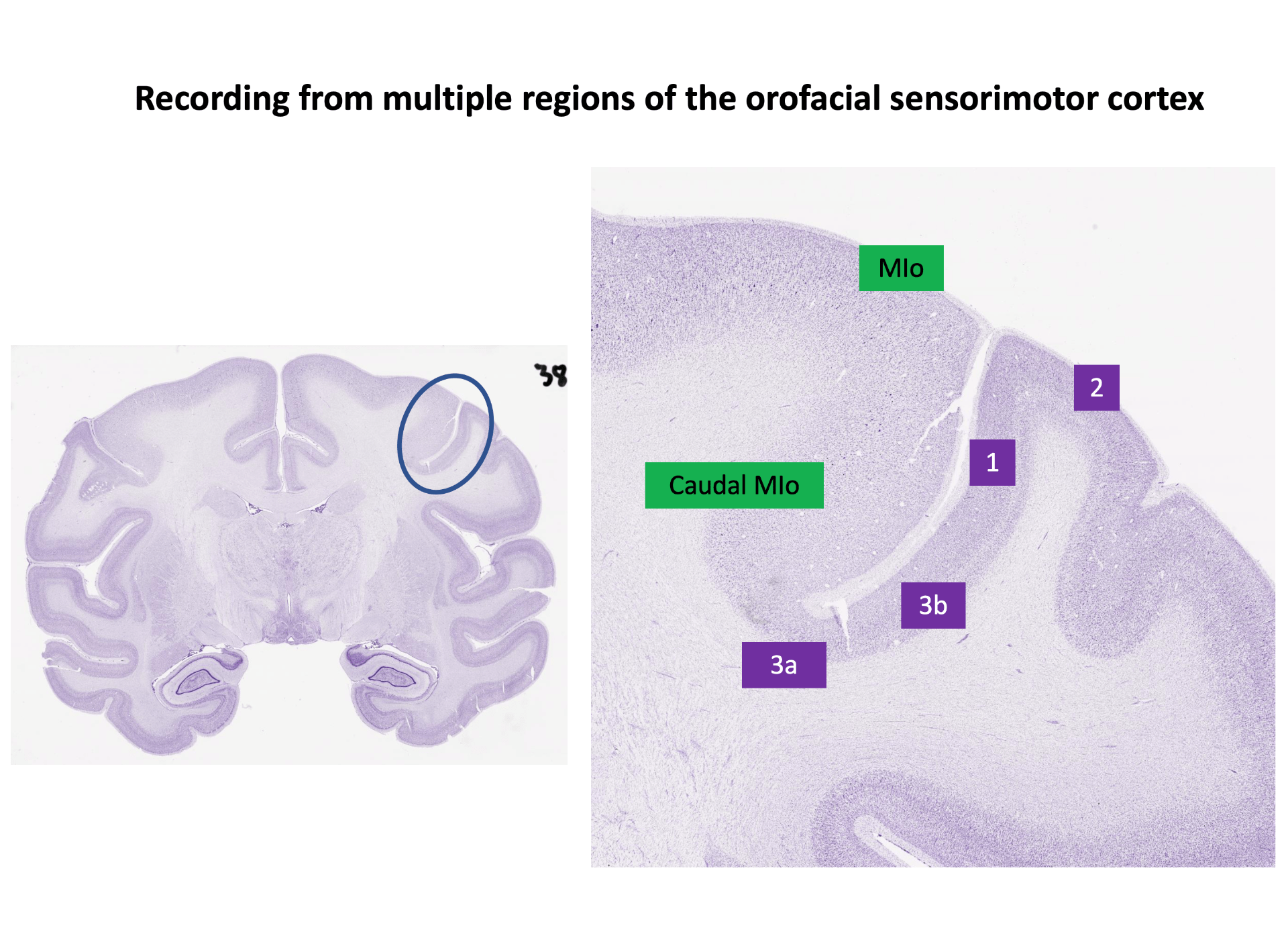
Our research focuses on the principles of cortical and biomechanical control of oral sensorimotor behavior and how these are affected by learning, aging, and disease. Our lab uses a multidisciplinary approach to understand the interplay between sensation and movement using converging evidence from psychophysics, biomechanics, neurophysiology, and computational modeling. Our methods include recording neural activity from chronically-implanted microelectrode arrays (Utah Arrays and Floating Microelectrode Arrays) in multiple regions of the cerebral cortex (rostral and caudal primary motor cortex, primary somatosensory cortex (1, 2, 3a, 3b), cortical masticatory area, ventrolateral frontal cortex) simultaneous with recording the kinematics of the tongue and the mandible using high-resolution biplanar radiography and applying computational models to understand the principles of sensorimotor control.
Projects
Neural Basis of Touch and Proprioception
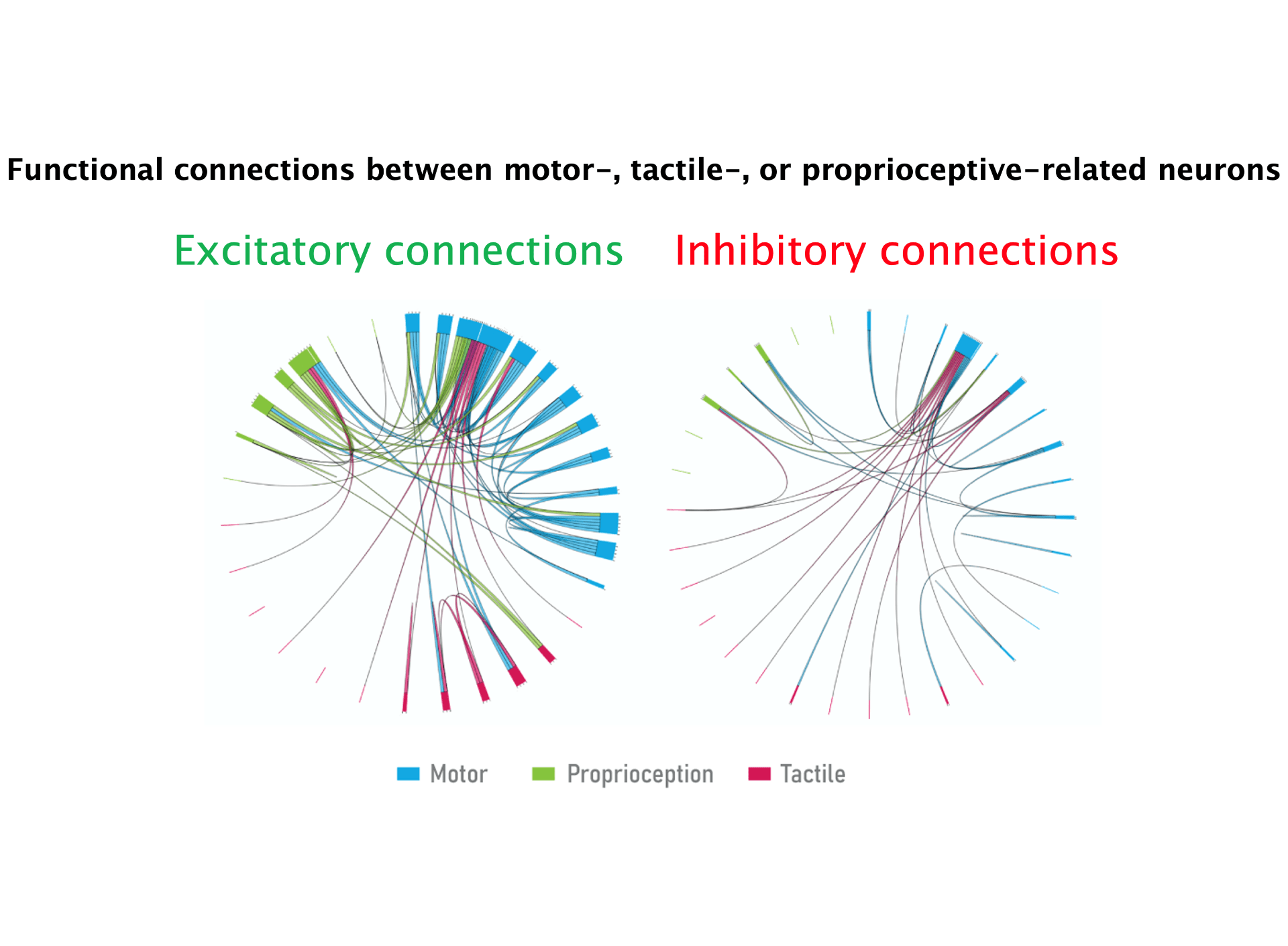
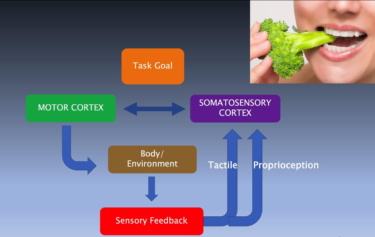
 Oral somatosensation plays a key role in vital functions of breathing, feeding, and speech such that altered sensation in pain disorders and sensory loss following neurological injuries have devastating effects on the quality of life. The project will provide novel insights on separate representations and integration of touch and proprioceptive sensations in the brain during natural feeding behavior and will lay the groundwork for future studies on oral somatosensation, pain mechanisms, and sensorimotor integration. It will inform the development of strategies for the evaluation and treatment of sensory impairments associated with dental implants and nerve injuries, trigeminal neuropathies, temporomandibular disorders, chronic pain, oromandibular dystonia, sensorimotor disorders, and for the restoration of sensory feedback for use in brain-machine interfaces.
Oral somatosensation plays a key role in vital functions of breathing, feeding, and speech such that altered sensation in pain disorders and sensory loss following neurological injuries have devastating effects on the quality of life. The project will provide novel insights on separate representations and integration of touch and proprioceptive sensations in the brain during natural feeding behavior and will lay the groundwork for future studies on oral somatosensation, pain mechanisms, and sensorimotor integration. It will inform the development of strategies for the evaluation and treatment of sensory impairments associated with dental implants and nerve injuries, trigeminal neuropathies, temporomandibular disorders, chronic pain, oromandibular dystonia, sensorimotor disorders, and for the restoration of sensory feedback for use in brain-machine interfaces.
Natural Aging vs Alzheimer’s Disease
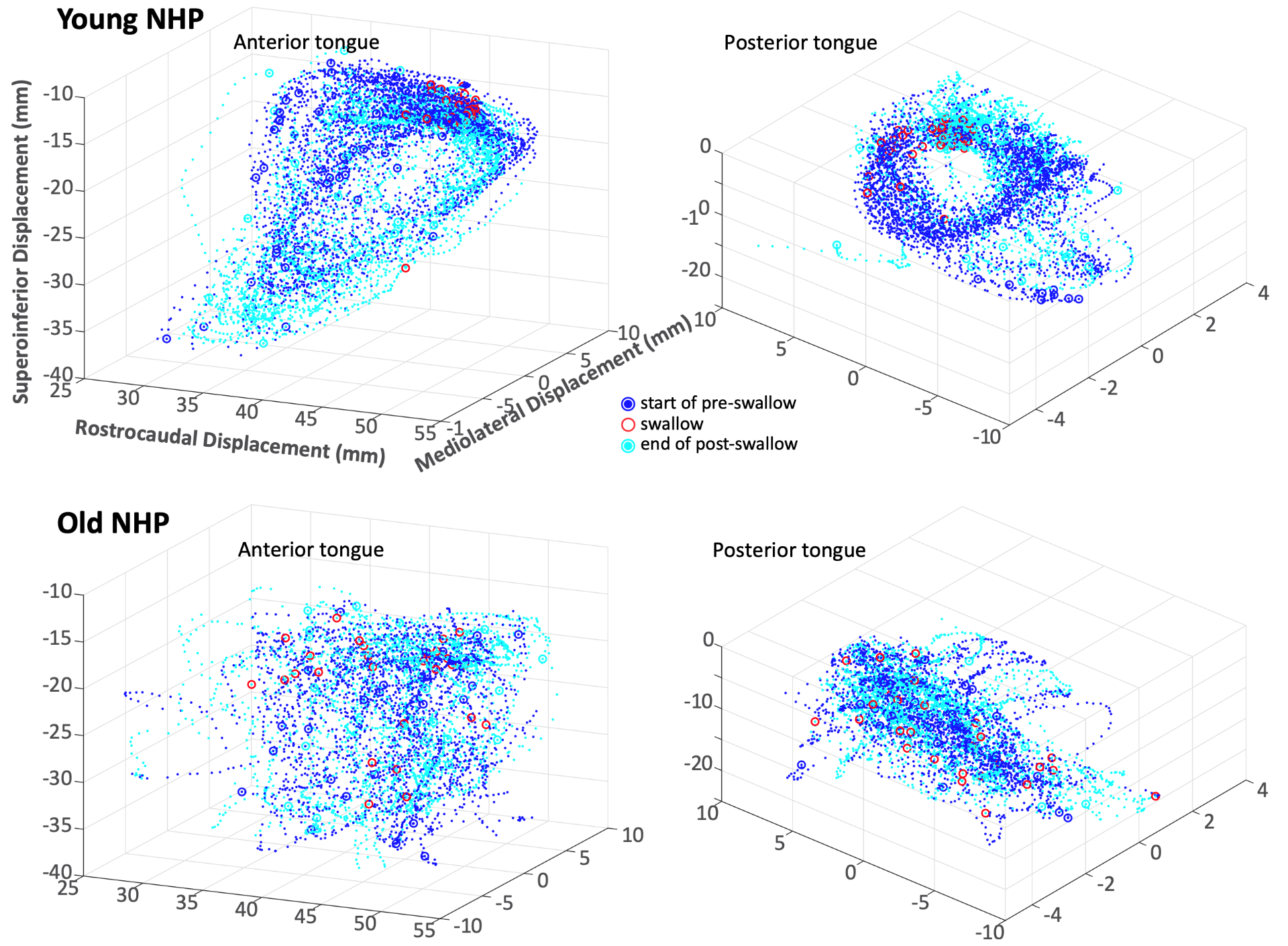
 The link between Alzheimer’s disease (AD) and age-related oral health problems is still poorly understood because changes in the cortical and biomechanical control of oromotor behavior in healthy aging and pathological aging have not been investigated. The proposed research is relevant to public health because disambiguating changes in the orofacial sensorimotor-cognitive neuronal network in healthy aging from pathological aging is expected to increase the understanding of potential contributing factors to the onset and progression of AD and the cortical areas that are vulnerable to AD. Thus, the proposed research is relevant to NIH’s mission of earlier identification of individuals who may be at risk for developing late-onset AD or age-related dementias and to find potential avenues for the evaluation of pharmacological or cortical therapies for AD.
The link between Alzheimer’s disease (AD) and age-related oral health problems is still poorly understood because changes in the cortical and biomechanical control of oromotor behavior in healthy aging and pathological aging have not been investigated. The proposed research is relevant to public health because disambiguating changes in the orofacial sensorimotor-cognitive neuronal network in healthy aging from pathological aging is expected to increase the understanding of potential contributing factors to the onset and progression of AD and the cortical areas that are vulnerable to AD. Thus, the proposed research is relevant to NIH’s mission of earlier identification of individuals who may be at risk for developing late-onset AD or age-related dementias and to find potential avenues for the evaluation of pharmacological or cortical therapies for AD.
Tools


Gallery
;